8-K
false000185027000018502702025-07-082025-07-08
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
|
Date of Report (Date of earliest event reported): July 08, 2025 |
PROKIDNEY CORP.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
Delaware |
001-40560 |
98-1586514 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
|
2000 Frontis Plaza Blvd. Suite 250 |
|
Winston-Salem, North Carolina |
|
27103 |
(Address of Principal Executive Offices) |
|
(Zip Code) |
|
Registrant’s Telephone Number, Including Area Code: 336 999-7019 |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class
|
|
Trading
Symbol(s) |
|
Name of each exchange on which registered
|
Class A common stock, $0.0001 par value per share |
|
PROK |
|
The Nasdaq Stock Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Item 7.01 Regulation FD Disclosure.
On July 8, 2025, ProKidney Corp. (the "Company") issued a press release to announce topline results for its REGEN-007 study. REGEN-007 is an ongoing Phase 2, prospective, randomized, open-label, repeat dose, two group, safety and efficacy study of rilparencel in subjects with type 1 or 2 diabetes and CKD. A copy of the press release is furnished as Exhibit 99.1
In addition, the Company has updated its investor presentation (the "Presentation"), which its senior management intends to use from time to time when interacting with investors and analysts, among others. The Presentation is available on the Company’s website at https://investors.prokidney.com/news-events/events-and-presentations. The Presentation is also attached hereto as Exhibit 99.2.
The information in this Current Report on Form 8-K (including Exhibits 99.1 and 99.2) is being furnished, not filed, pursuant to Regulation FD. Accordingly, the information in Items 7.01 and 9.01 of this report will not be incorporated by reference into any registration statement filed by the Company under the Securities Act of 1933, as amended, unless specifically identified therein as being incorporated therein by reference. The furnishing of the information in this report is not intended to, and does not, constitute a determination or admission by the Company that the information in this report is material or complete, or that investors should consider this information before making an investment decision with respect to any security of the Company or any of its affiliates.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
PROKIDNEY CORP. |
|
|
|
|
Date: |
July 8, 2025 |
By: |
/s/ Todd Girolamo |
|
|
|
Todd Girolamo
Chief Legal Officer |
EX-99.1
Exhibit 99.1

ProKidney Reports Statistically and Clinically Significant Topline Results for the Phase 2 REGEN-007 Trial Evaluating Rilparencel in Patients with Chronic Kidney Disease and Diabetes
•Full results from REGEN-007 are being held and will be submitted to the American Society of Nephrology 2025 Kidney Week as a late-breaking clinical trial
•In Group 1 (n=24), kidney function stabilized in patients randomized to receive two rilparencel injections (one in each kidney). The annual decline in eGFR slope improved by 78% from -5.8 mL/min/1.73m2 in the pre-injection period to -1.3 mL/min/1.73m2 in the period following the last rilparencel injection. This 4.6 mL/min/1.73m2 per year difference was statistically significant (p<0.001) and clinically meaningful
•In Group 2 (n=25), patients were randomized to receive a single rilparencel injection followed by a second injection only if kidney function worsened and a re-dosing trigger was met. The annual decline in eGFR slope improved by 50% from -3.4 mL/min/1.73m2 in the pre-injection period to -1.7 mL/min/1.73m2 in the period following the last rilparencel injection. This 1.7 mL/min/1.73m2 per year difference was not statistically significant (p=0.085) but suggests evidence of a dose response
•No rilparencel-related serious adverse events were observed; the safety profile was consistent with previously reported study results and comparable to a kidney biopsy
•FDA Type B meeting set for this summer to confirm ProKidney’s approach to using eGFR slope as the surrogate endpoint in the ongoing Phase 3 PROACT 1 study for accelerated approval
WINSTON-SALEM, N.C., July 8, 2025 – ProKidney Corp. (Nasdaq: PROK) (“ProKidney” or the “Company"), a leading late clinical-stage cellular therapeutics company focused on chronic kidney disease (CKD), today reported statistically significant and clinically meaningful positive topline results from the full Group 1 modified intent-to-treat (mITT) population of the Phase 2 REGEN-007 trial evaluating rilparencel in patients with CKD and diabetes. Rilparencel is an autologous cellular therapy that has received Regenerative Medicine Advanced Therapy (RMAT) designation from the U.S. Food & Drug Administration (FDA) and is currently being evaluated in the ongoing Phase 3 REGEN-006 (PROACT 1) trial to demonstrate the therapy’s potential to preserve kidney function in patients with advanced CKD and type 2 diabetes.
“We are very encouraged by the REGEN-007 topline results that demonstrated a robust improvement in eGFR slope following treatment with rilparencel in Group 1 as well as evidence of a dose response in Group 2. These data bolster our confidence in the design of our ongoing Phase 3 PROACT 1 study given the similarity between the dosing regimen in REGEN-007 Group 1 and PROACT 1. It is also worth noting that 15 of the 24 patients in Group 1 (63%) met key Phase 3 PROACT 1 inclusion criteria, and similar efficacy results were observed in this subgroup compared to the full Group 1 results. We plan to submit the full results from REGEN-007 to ASN’s 2025 Kidney Week as a late-breaking clinical trial and are excited to share more details at that time with investors and the medical community,” said Bruce Culleton, M.D., CEO of ProKidney. “We also look forward to our upcoming FDA Type B meeting in the coming weeks to confirm our approach to eGFR slope as a surrogate endpoint for accelerated approval. This meeting
represents an important step toward our goal of expediting rilparencel’s potential path to market in the U.S. where there remains a significant unmet clinical need in patients with advanced CKD and diabetes.”
Phase 2 REGEN-007 Overview and Topline Results
REGEN-007 is a multi-center Phase 2 open-label 1:1 randomized two-arm trial in patients with diabetes, CKD, and an estimated glomerular filtration rate (eGFR) of 20-50 mL/min/1.73m². At randomization, patients were assigned to one of two treatment groups using different dosing regimens. Group 1 replicated the dosing schedule of the ongoing Phase 3 PROACT 1 study in which patients received two scheduled rilparencel injections (one in each kidney), approximately three months apart. Group 2 tested an exploratory dosing regimen to investigate whether disease progression triggers, rather than a time-based trigger, could optimize multiple administrations of rilparencel. In Group 2, patients received a single rilparencel injection in one kidney and a second injection in the contralateral kidney only if triggered by a sustained eGFR decline from baseline of ≥ 20%, and/or an increase in the urine albumin to creatinine ratio (UACR) from baseline of ≥ 30% and ≥ 30 mg/g.
The prespecified primary endpoint for REGEN-007 is the difference in annual eGFR slope (calculated using a linear mixed effects model) in the pre-injection period versus the period following the last rilparencel injection. The pre-injection period included all historical eGFR values collected up to 24 months before the screening visit as well as the on-study central laboratory eGFR results prior to first rilparencel injection. The period following the last injection included eGFR values from the last rilparencel injection to the end of study (EOS) visit. Median follow-up after the last injection was approximately 18 months in both Group 1 and Group 2.
Fifty-three patients were randomized in the study, of whom 49 patients (mITT population) received at least one rilparencel injection. Four patients did not receive any rilparencel injections. The majority of patients were male (69%), and the mean age was 60 years. At baseline, 38 of 49 patients (78%) had type 2 diabetes mellitus and 11 (22%) had type 1 diabetes. Thirty-nine (80%) patients were receiving an angiotensin-converting enzyme inhibitor (ACEi) or an angiotensin II receptor blocker (ARB), and 18 (37%) were receiving a sodium-glucose cotransporter-2 inhibitor (SGLT2i). At baseline, the mean (SD) eGFR was 33±10 mL/min/1.73m2. Notably, the median UACR was higher in Group 1 (792 mg/g) compared to Group 2 (229 mg/g).

In Group 1 (n=24), kidney function stabilized after receiving rilparencel. The annual decline in eGFR slope improved by 78% from -5.8 mL/min/1.73m2 in the pre-injection period to -1.3 mL/min/1.73m2 in the period following the last rilparencel injection. This 4.6 mL/min/1.73m2 per year difference1 was statistically significant (p<0.001) and clinically meaningful. Of the 24 patients in Group 1, 15 (63%) met key Phase 3 PROACT 1 inclusion criteria, and similar efficacy results were observed in this subgroup compared to the full Group 1 results. As a reminder, the Phase 3 PROACT 1 protocol was amended in 1H 2024 after a similar eGFR efficacy signal was observed in the Phase 2 RMCL-002 study subgroup analysis (n=23) of high-UACR, Stage 4 CKD patients with type 2 diabetes.
1 Difference in values is due to rounding.
In Group 2 (n=25), the annual change in kidney function as measured by eGFR slope was -3.4 mL/min/1.73m2 in the pre-injection period versus -1.7 mL/min/1.73m2 in the period following the last rilparencel injection, resulting in an improvement of 50%, or 1.7 mL/min/1.73m2 per year. This difference was not statistically significant (p=0.085) but suggests evidence of a dose response. Out of the 25 patients in Group 2, 15 (60%) met the re-dosing trigger and received a second rilparencel injection. The median time between the first and second injections in these 15 patients was approximately 11 months.
No rilparencel-related serious adverse events were observed across all patients in the study who received at least one rilparencel injection (n=49). The safety profile was consistent with previously reported study results and comparable to a kidney biopsy.
Full results from REGEN-007 are being held and will be submitted to the American Society of Nephrology (ASN) 2025 Kidney Week as a late-breaking clinical trial.
Phase 3 PROACT 1 Regulatory Progress
As previously communicated, the FDA confirmed during a Type B meeting in Q4 2024 that the accelerated approval pathway is available for rilparencel if an acceptable surrogate endpoint, such as eGFR slope, is used. ProKidney has an upcoming FDA Type B meeting this summer to confirm the approach to using eGFR slope as the surrogate endpoint for accelerated approval. Additional details are expected in mid-2025.
About Chronic Kidney Disease
CKD is a progressive condition characterized by the gradual decline of kidney function, which can ultimately lead to end-stage kidney disease (ESKD) requiring dialysis or transplantation. An estimated 37 million adults in the U.S. have CKD, though many remain undiagnosed in the early stages. Diabetes is the leading cause of CKD, and individuals with both conditions face significantly elevated risks of cardiovascular events, hospitalization, and mortality. ProKidney is developing rilparencel for patients with Stage 3b/4 CKD and diabetes, a population that includes 1 to 2 million people in the U.S. While current treatment options aim to slow disease progression, there remains a substantial unmet need for therapies that can stabilize kidney function and delay or prevent the need for dialysis in patients with advanced CKD.
About the Phase 3 REGEN-006 (PROACT 1) Clinical Trial
REGEN-006 is an ongoing Phase 3, randomized, blinded, sham controlled safety and efficacy study of rilparencel in subjects with advanced CKD and type 2 diabetes. The study protocol was amended in 1H 2024 to focus on a subset of patients with Stage 4 CKD (eGFR 20-30ml min/1.73m2) and late Stage 3b CKD (eGFR 30-35ml min/1.73m2) with accompanying albuminuria (UACR less than 5,000 mg/g for patients with eGFR 20-30ml min/1.73m2 and 300-5,000 mg/g for patients with eGFR 30-35ml min/1.73m2). The total planned enrollment is approximately 685 subjects. Subjects are randomized (1:1) to the treatment group and the sham control group prior to kidney biopsy or a sham biopsy procedure, respectively. The primary objective is to assess the efficacy of up to two rilparencel injections (one in each kidney) using a minimally invasive percutaneous approach. The primary composite endpoint is the time from first injection to the earliest of: at least 40% reduction in eGFR; eGFR <15 mL/min/1.73m², and/or chronic dialysis, and/or renal transplant; or renal or cardiovascular death.
About ProKidney Corp.
ProKidney, a pioneer in the treatment of chronic kidney disease through innovations in cellular therapy, was founded in 2015 after a decade of research. ProKidney’s lead product candidate, rilparencel (also
known as REACT®), is a first-in-class, patented, proprietary autologous cellular therapy being evaluated for its potential to preserve kidney function in diabetic patients at high risk of kidney failure. Rilparencel has received RMAT designation from the FDA. For more information, please visit www.prokidney.com.
Forward-Looking Statements
This press release includes “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. ProKidney’s actual results may differ from its expectations, estimates and projections and consequently, you should not rely on these forward-looking statements as predictions of future events. Words such as “expect,” “estimate,” “project,” “budget,” “forecast,” “anticipate,” “intend,” “plan,” “may,” “will,” “could,” “should,” “believes,” “predicts,” “potential,” “continue,” and similar expressions (or the negative versions of such words or expressions) are intended to identify such forward-looking statements. These forward-looking statements include, without limitation, the Company’s beliefs that the FDA agrees that the Company’s Phase 3 REGEN-006 (PROACT 1) trial could be sufficient to support a potential BLA submission and full regulatory approval and that the Company could consider using eGFR slope as a surrogate endpoint on an accelerated approval pathway for rilparencel, expectations with respect to financial results and expected cash runway, including the Company’s expectation that current cash will support operating plans into 2027, future performance, development and commercialization of products, if approved, the potential benefits and impact of the Company’s products, if approved, potential regulatory approvals, the size and potential growth of current or future markets for the Company’s products, if approved, the advancement of the Company’s development programs into and through the clinic and the expected timing for reporting data, the making of regulatory filings or achieving other milestones related to the Company’s product candidates, and the advancement and funding of the Company’s developmental programs, generally. Most of these factors are outside of the Company’s control and are difficult to predict. Factors that may cause such differences include, but are not limited to: disruptions to our business or that may otherwise materially harm our results of operations or financial condition as a result of our recent domestication to the United States; the inability to maintain the listing of the Company’s Class A common stock on Nasdaq; the inability of the Company’s Class A common stock to remain included in various indices and the potential negative impact on the trading price of the Class A common stock if excluded from such indices; the inability to implement business plans, forecasts, and other expectations or identify and realize additional opportunities, which may be affected by, among other things, competition and the ability of the Company to grow and manage growth profitably and retain its key employees; the risk of downturns and a changing regulatory landscape in the highly competitive biotechnology industry; the risk that results of the Company’s clinical trials may not support approval; the risk that the FDA could require additional studies before approving the Company’s drug candidates; the inability of the Company to raise financing in the future; the inability of the Company to obtain and maintain regulatory clearance or approval for its products, and any related restrictions and limitations of any cleared or approved product; the inability of the Company to identify, in-license or acquire additional technology; the inability of Company to compete with other companies currently marketing or engaged in the biologics market and in the area of treatment of kidney diseases; the size and growth potential of the markets for the Company’s products, if approved, and its ability to serve those markets, either alone or in partnership with others; the Company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; the Company’s financial performance; the Company’s intellectual property rights; uncertainties inherent in cell therapy research and development, including the actual time it takes to initiate and complete clinical studies and the timing and content of decisions made by regulatory authorities; the fact that interim results from our clinical programs may not be indicative of future results; the impact of geo-political conflict on the Company’s business; and other risks and uncertainties included under the heading “Risk Factors” in the Company’s most recent Annual Report on Form 10-K, subsequent Quarterly Reports on
Form 10-Q and other filings with the Securities and Exchange Commission. The Company cautions readers that the foregoing list of factors is not exclusive and cautions readers not to place undue reliance upon any forward-looking statements, which speak only as of the date made. The Company does not undertake or accept any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements to reflect any change in its expectations or any change in events, conditions or circumstances on which any such statement is based.
Investor Contacts:
ProKidney
Ethan Holdaway
Ethan.Holdaway@prokidney.com
LifeSci Advisors, LLC
Daniel Ferry
Daniel@lifesciadvisors.com

March 2024 July 2025 Corporate Presentation Exhibit 99.2

This presentation includes “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995. ProKidney’s actual results may differ from its expectations, estimates and projections and consequently, you should not rely on these forward-looking statements as predictions of future events. Words such as “expect,” “estimate,” “project,” “budget,” “forecast,” “anticipate,” “intend,” “plan,” “may,” “will,” “could,” “should,” “believes,” “predicts,” “potential,” “continue,” and similar expressions (or the negative versions of such words or expressions) are intended to identify such forward-looking statements. These forward-looking statements include, without limitation, the Company’s beliefs that the FDA agrees that the Company’s Phase 3 REGEN-006 (PROACT 1) trial could be sufficient to support a potential BLA submission and full regulatory approval and that the Company could consider using eGFR slope as a surrogate endpoint on an accelerated approval pathway for rilparencel, expectations with respect to financial results and expected cash runway, including the Company’s expectation that current cash will support operating plans into mid-2027, future performance, development and commercialization of products, if approved, the potential benefits and impact of the Company’s products, if approved, potential regulatory approvals, the size and potential growth of current or future markets for the Company’s products, if approved, the advancement of the Company’s development programs into and through the clinic and the expected timing for reporting data, the making of regulatory filings or achieving other milestones related to the Company’s product candidates, and the advancement and funding of the Company’s developmental programs, generally. Most of these factors are outside of the Company’s control and are difficult to predict. Factors that may cause such differences include, but are not limited to: disruptions to our business that may otherwise materially harm our results of operations or financial condition as a result of our recent domestication to the United States; the inability to maintain the listing of the Company’s Class A common stock on Nasdaq; the inability of the Company’s Class A common stock to remain included in various indices and the potential negative impact on the trading price of the Class A common stock if excluded from such indices; the inability to implement business plans, forecasts, and other expectations or identify and realize additional opportunities, which may be affected by, among other things, competition and the ability of the Company to grow and manage growth profitably and retain its key employees; the risk of downturns and a changing regulatory landscape in the highly competitive biotechnology industry; the risk that results of the Company’s clinical trials may not support approval; the risk that the FDA could require additional studies before approving the Company’s drug candidates; the inability of the Company to raise financing in the future; the inability of the Company to obtain and maintain regulatory clearance or approval for its products, and any related restrictions and limitations of any cleared or approved product; the inability of the Company to identify, in-license or acquire additional technology; the inability of Company to compete with other companies currently marketing or engaged in the biologics market and in the area of treatment of kidney diseases; the size and growth potential of the markets for the Company’s products, if approved, and its ability to serve those markets, either alone or in partnership with others; the Company’s estimates regarding expenses, future revenue, capital requirements and needs for additional financing; the Company’s financial performance; the Company’s intellectual property rights; uncertainties inherent in cell therapy research and development, including the actual time it takes to initiate and complete clinical studies and the timing and content of decisions made by regulatory authorities; the fact that interim results from our clinical programs may not be indicative of future results; the impact of geo-political conflict on the Company’s business; and other risks and uncertainties included under the heading “Risk Factors” in the Company’s most recent Annual Report on Form 10-K, subsequent Quarterly Reports on Form 10-Q and other filings with the Securities and Exchange Commission. The Company cautions readers that the foregoing list of factors is not exclusive and cautions readers not to place undue reliance upon any forward-looking statements, which speak only as of the date made. The Company does not undertake or accept any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements to reflect any change in its expectations or any change in events, conditions or circumstances on which any such statement is based. This presentation shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities laws of any such state or jurisdiction. Forward-looking Statements

Renal Autologous Cell Therapy: Rilparencel (REACT®) proprietary autologous cellular therapy being evaluated to �preserve kidney function in patients with diabetes and advanced chronic kidney disease Disrupting the CKD Treatment Landscape

Recent Developments An Introduction to ProKidney CKD = chronic kidney disease RMAT = regenerative medicine advanced therapy Preserve kidney function in advanced CKD patients Preserve kidney function in patients with type 2 diabetes and advanced chronic kidney disease who are faced with limited options for care beyond transplantation or dialysis A proprietary autologous cellular therapy with RMAT designation Currently in pivotal Phase 3 clinical development with REGEN-006 (PROACT 1) Supported by three Phase 2 clinical trials in advanced CKD patient populations Leadership Team with Clinical Development & Regulatory Experience Together the team brings over 150 years cumulative experience in the discovery, development, manufacturing and commercialization of biotechnology, pharmaceutical, and device products Key Developments Across Clinical and Regulatory Objectives In July 2025, announced positive topline results for the Phase 2 REGEN-007 trial In a Q4 2024 Type B meeting, the FDA confirmed the accelerated approval pathway is available for rilparencel if an acceptable surrogate endpoint, such as eGFR slope, is used; additional details expected in mid-2025 Goal Rilparencel Leadership
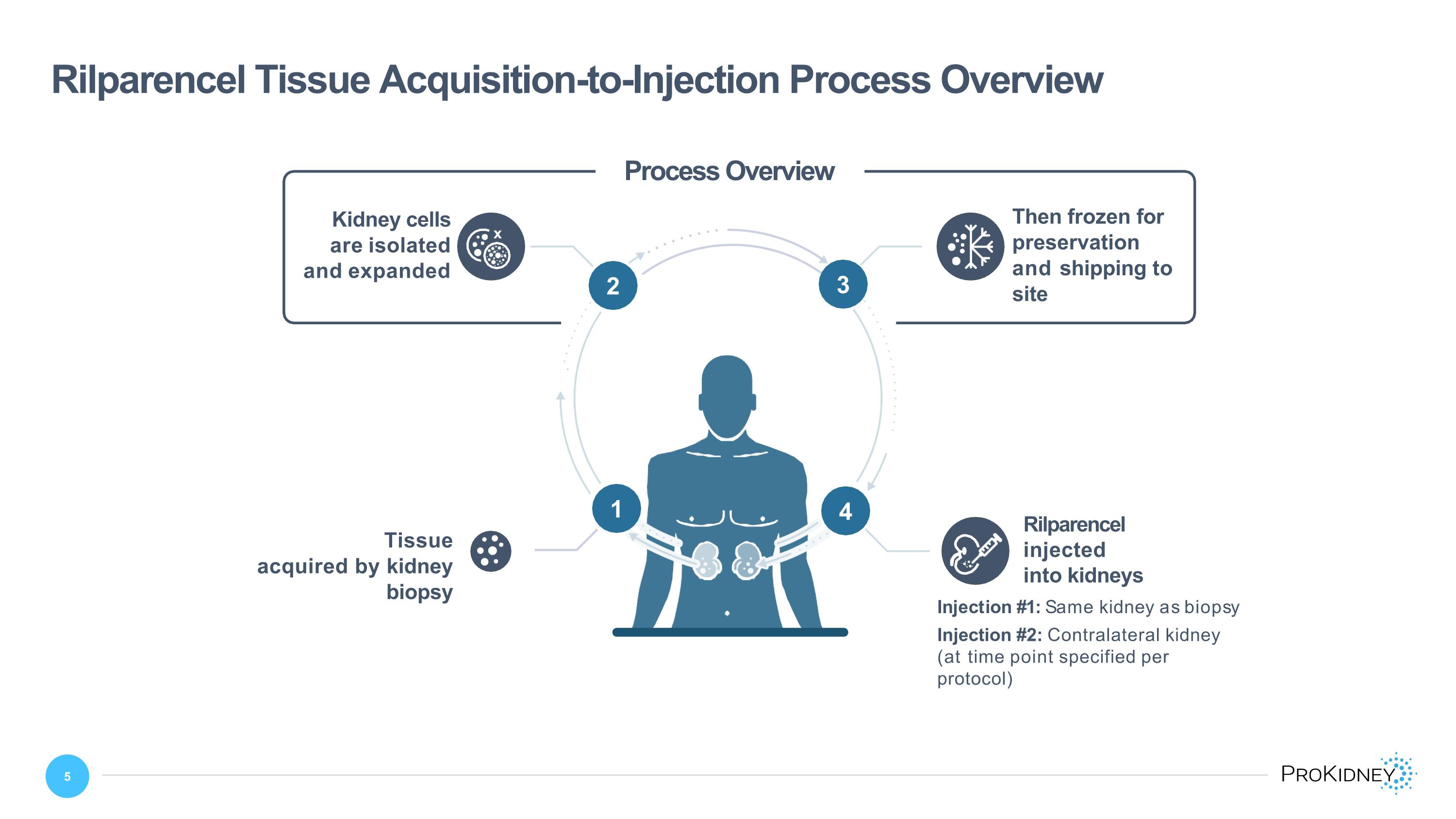
Rilparencel Tissue Acquisition-to-Injection Process Overview 4 2 3 x Then frozen for preservation and shipping to site Kidney cells are isolated and expanded Tissue acquired by kidney biopsy Rilparencel injected into kidneys 1 Process Overview Injection #1: Same kidney as biopsy Injection #2: Contralateral kidney (at time point specified per protocol) 13 2 3 4
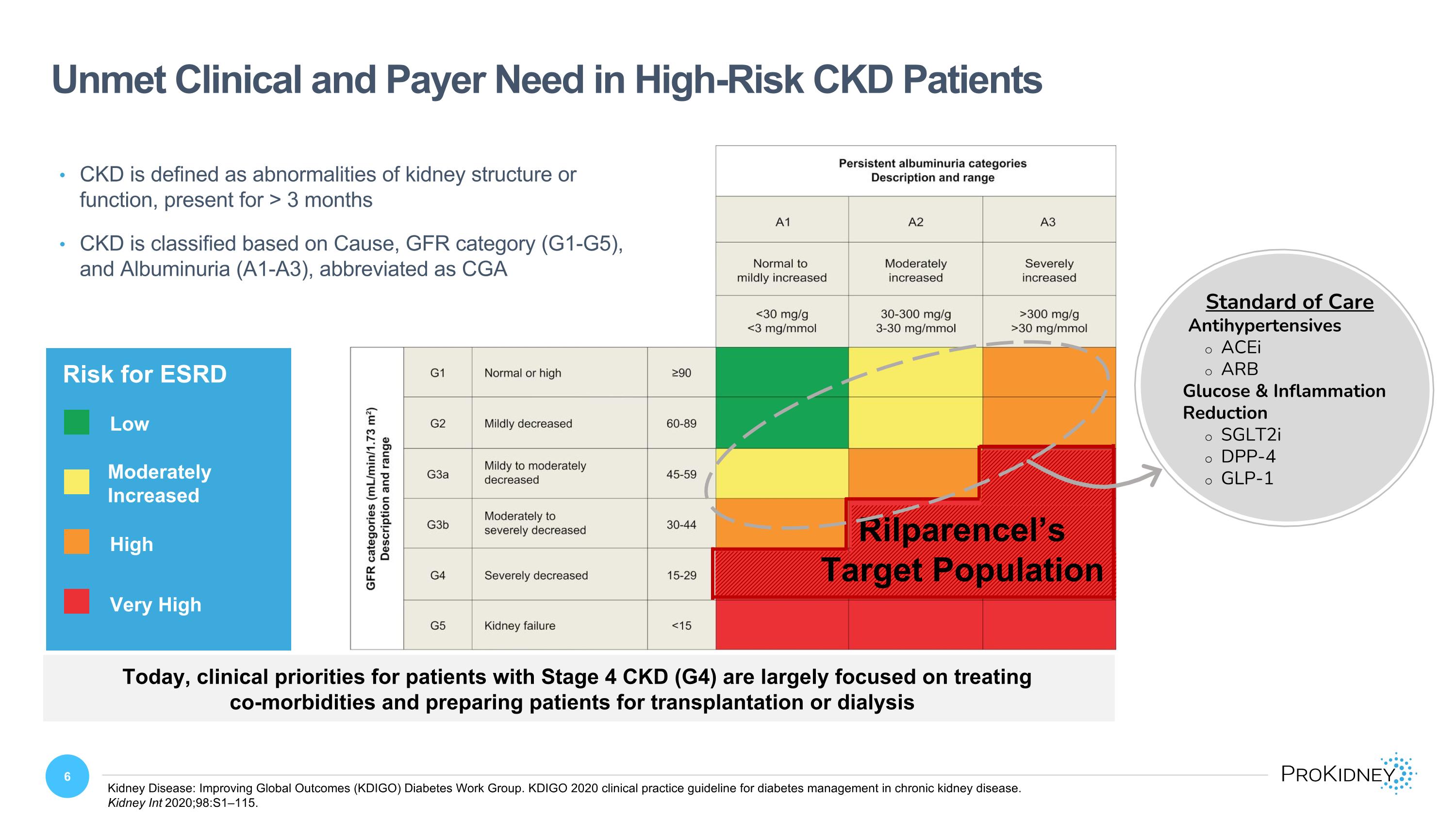
Unmet Clinical and Payer Need in High-Risk CKD Patients Kidney Disease: Improving Global Outcomes (KDIGO) Diabetes Work Group. KDIGO 2020 clinical practice guideline for diabetes management in chronic kidney disease. �Kidney Int 2020;98:S1–115. Moderately Increased High Very High Rilparencel’s Target Population Risk for ESRD Low Moderately Increased High Very High CKD is defined as abnormalities of kidney structure or function, present for > 3 months CKD is classified based on Cause, GFR category (G1-G5), and Albuminuria (A1-A3), abbreviated as CGA Standard of Care Antihypertensives ACEi ARB Glucose & Inflammation Reduction SGLT2i DPP-4 GLP-1 Today, clinical priorities for patients with Stage 4 CKD (G4) are largely focused on treating co-morbidities and preparing patients for transplantation or dialysis Persistent albuminuria categories Description and range A1 Normal to mildly increased <30 mg/g <3 mg/mmol A2 Moderately increased 30-300 mg/g 3-30 mg/mmol A3 Severely increased >300 mg/g >30 mg/mmol GFR categories (mL/min/1.73m2) Description and range G1 Normal or high ≥90 G2 Mildly decreased 60-89 G3a Mildy to moderately decreased 45-59 G3b Moderately to severely decreased 30-44 G4 Severely decreased 15-29 G5 Kidney failure <15

Study Active Product Subjects with Stage 4 CKD Canagliflozin and Renal Outcomes in Type 2 Diabetes and Nephropathy1 Canagliflozin (SGLT2 inhibitor) 0% Dapagliflozin in Patients with CKD2 Dapagliflozin (SGLT2 inhibitor) 14% Empagliflozin in Patients with CKD3 Empagliflozin (SGLT2 inhibitor) 34% Effect of Finerenone on Cardiovascular and Kidney Outcomes in Patients with Type 2 Diabetes and CKD4 Finerenone (Selective MRA) 7% Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes5 Semaglutide (GLP-1RA) 11% Therapeutic Options that Delay the Need for Dialysis in Patients with �Stage 4 Chronic Kidney Disease are Limited All recent landmark clinical trials in CKD primarily focus on Stage 2 and 3 CKD Perkovic V et al. N Eng J Med 2019 Heerspink H et al. N Engl J Med 2020 Herrington et al. N Engl J Med 2023 Agarwal. R et al. Eur Heart J. 2022; �Sarafidis. P et al. Clin J Am Soc Nephrol 2023 Perkovic V et al. N Engl J Medicine 2024
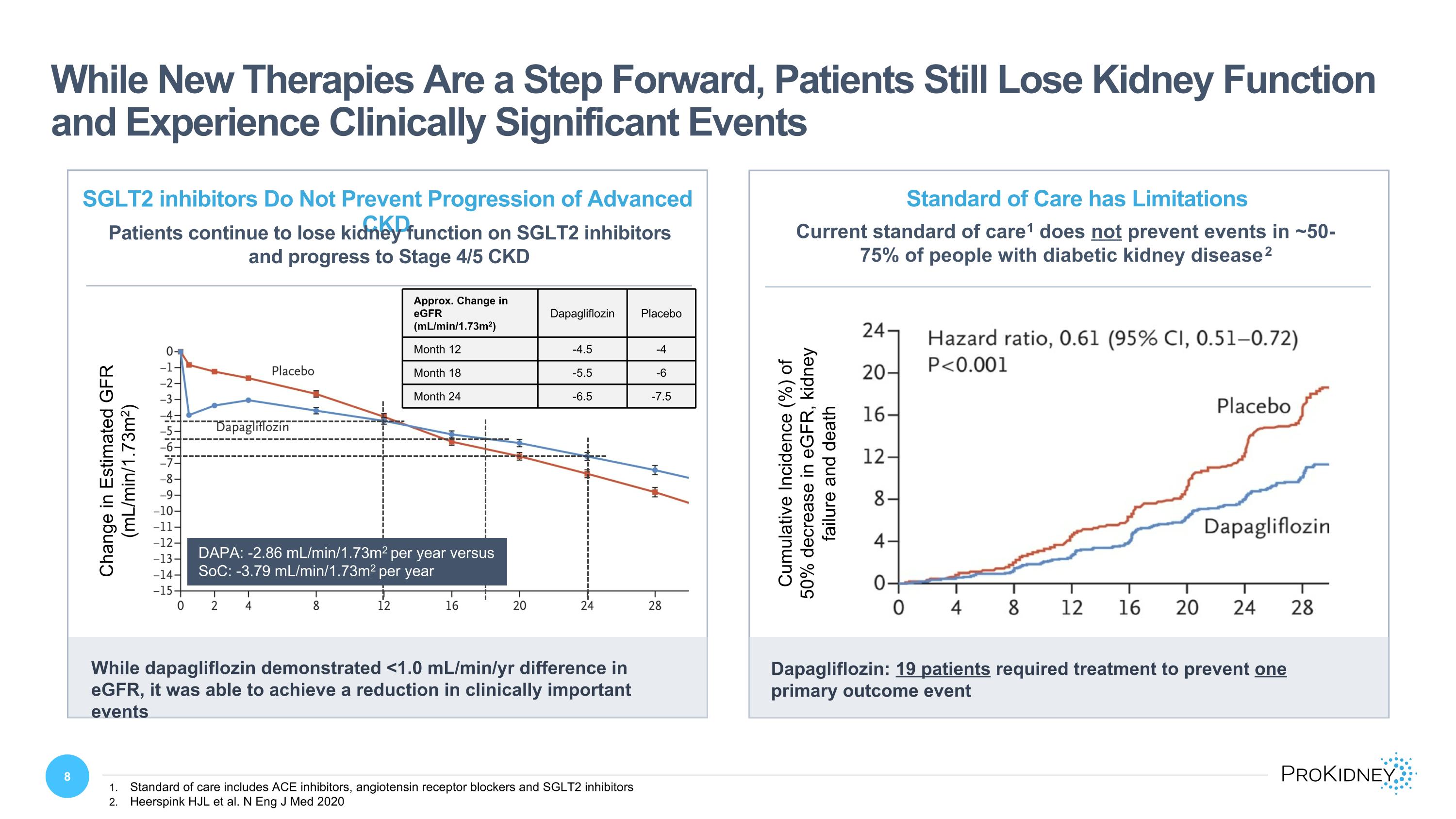
While New Therapies Are a Step Forward, Patients Still Lose Kidney Function and Experience Clinically Significant Events Standard of care includes ACE inhibitors, angiotensin receptor blockers and SGLT2 inhibitors Heerspink HJL et al. N Eng J Med 2020 Current standard of care1 does not prevent events in ~50-75% of people with diabetic kidney disease2 Standard of Care has Limitations Cumulative Incidence (%) of 50% decrease in eGFR, kidney failure and death Dapagliflozin: 19 patients required treatment to prevent one primary outcome event SGLT2 inhibitors Do Not Prevent Progression of Advanced CKD Patients continue to lose kidney function on SGLT2 inhibitors and progress to Stage 4/5 CKD Change in Estimated GFR (mL/min/1.73m2) While dapagliflozin demonstrated <1.0 mL/min/yr difference in eGFR, it was able to achieve a reduction in clinically important events Approx. Change in eGFR (mL/min/1.73m2) Dapagliflozin Placebo Month 12 -4.5 -4 Month 18 -5.5 -6 Month 24 -6.5 -7.5 DAPA: -2.86 mL/min/1.73m2 per year versus �SoC: -3.79 mL/min/1.73m2 per year
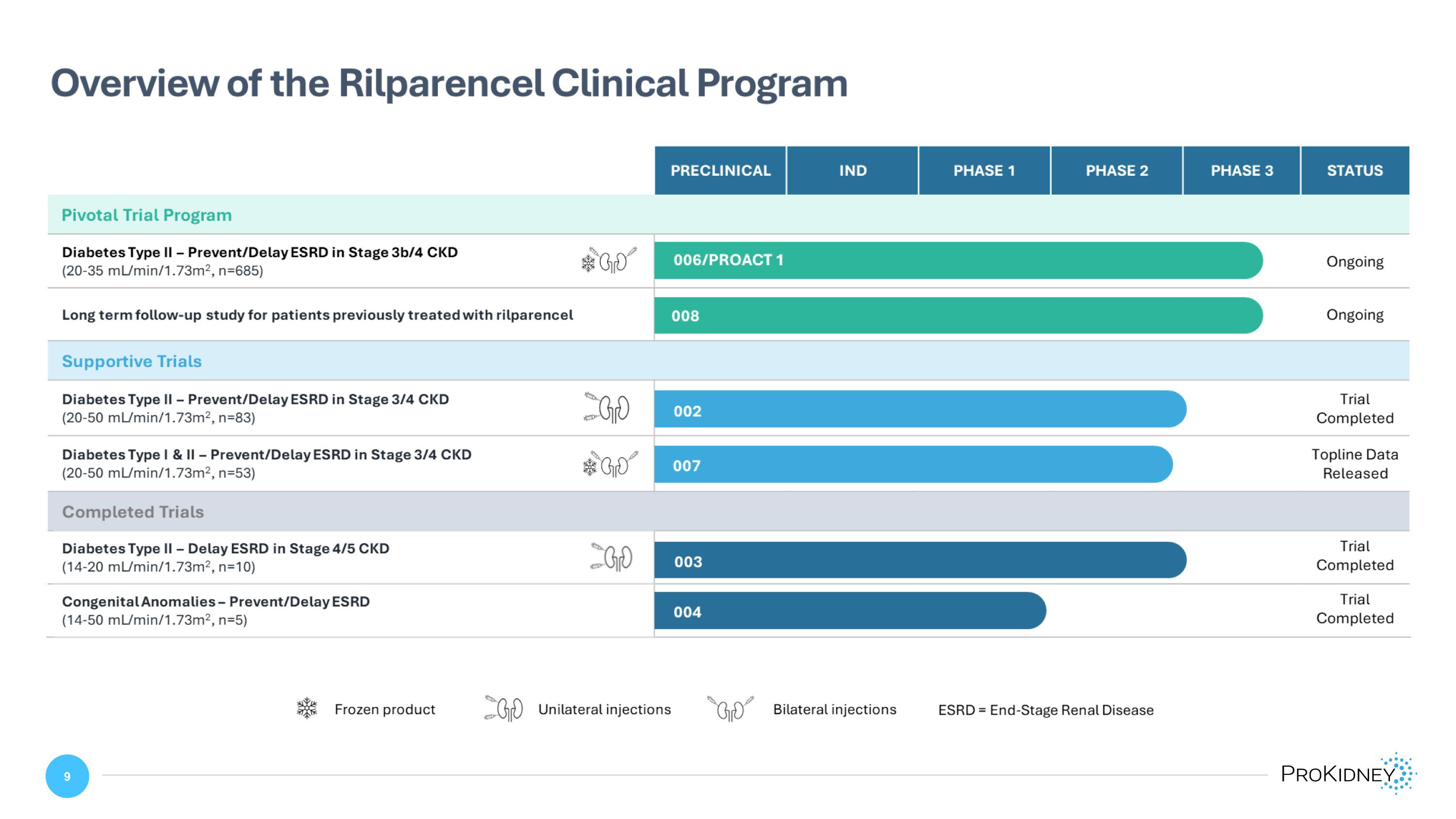
Overview of the Rilparencel Clinical Program PRECLINICAL IND PHASE 1 PHASE 2 PHASE 3 STATUS Pivotal Trial Program Diabetes Type II – Prevent/Delay ESRD in Stage 3b/4 CKD (20-35 mL/min/1.73m2, n=685) 006/PROACT 1 Ongoing Long term follow-up study for patients previously treated with rilparencel 008 Ongoing Supportive Trials Diabetes Type II – Prevent/Delay ESRD in Stage 3/4 CKD (20-50 mL/min/1.73m2, n=83) 002 Trial Completed Diabetes Type I & II – Prevent/Delay ESRD in Stage 3/4 CKD�(20-50 mL/min/1.73m2, n=53) 007 Topline Data Released Completed Trials Diabetes Type II – Delay ESRD in Stage 4/5 CKD (14-20 mL/min/1.73m2, n=10) 003 Trial Completed Congenital Anomalies – Prevent/Delay ESRD (14-50 mL/min/1.73m2, n=5) 004 Trial Completed Frozen product Unilateral injections Bilateral injections ESRD = End-Stage Renal Disease

Advancing a Comprehensive Clinical Plan RMCL-002 Phase 2 Trial; full results published in May 2024 Open-label safety & efficacy trial of rilparencel in patients with Stage 3/4 CKD (eGFR 20-50) and type 2 diabetes Results indicated the potential to preserve kidney function for up to 30 months in several patient groups REGEN-006 (PROACT 1) Phase 3 Trial in patients with Stage 3b/4 CKD and type 2 diabetes In a Q4 2024 Type B meeting, FDA confirmed that PROACT 1 could be sufficient to support a full U.S. rilparencel approval FDA also confirmed that the accelerated approval pathway is available for rilparencel if an acceptable surrogate endpoint, such as eGFR slope, is used 2024 REGEN-007 Phase 2 Trial; positive topline results announced in July 2025 Open-label safety & efficacy trial of rilparencel in patients with Stage 3/4 CKD (eGFR 20-50) and diabetes Patients in Group 1 (n=24) met the primary endpoint demonstrating kidney function stabilization Safety profile was consistent with previously reported study results and comparable to a kidney biopsy Full results will be submitted as a late-breaking clinical trial to the American Society of Nephrology (ASN) Kidney Week REGEN-006 (PROACT 1) Phase 3 Trial in patients with Stage 3b/4 CKD and type 2 diabetes Upcoming FDA Type B meeting in summer 2025 to confirm the approach using eGFR slope as the surrogate endpoint for the accelerated approval path for rilparencel Additional details expected in mid-2025 Update on rilparencel mechanism of action in 2H 2025 2025 and beyond
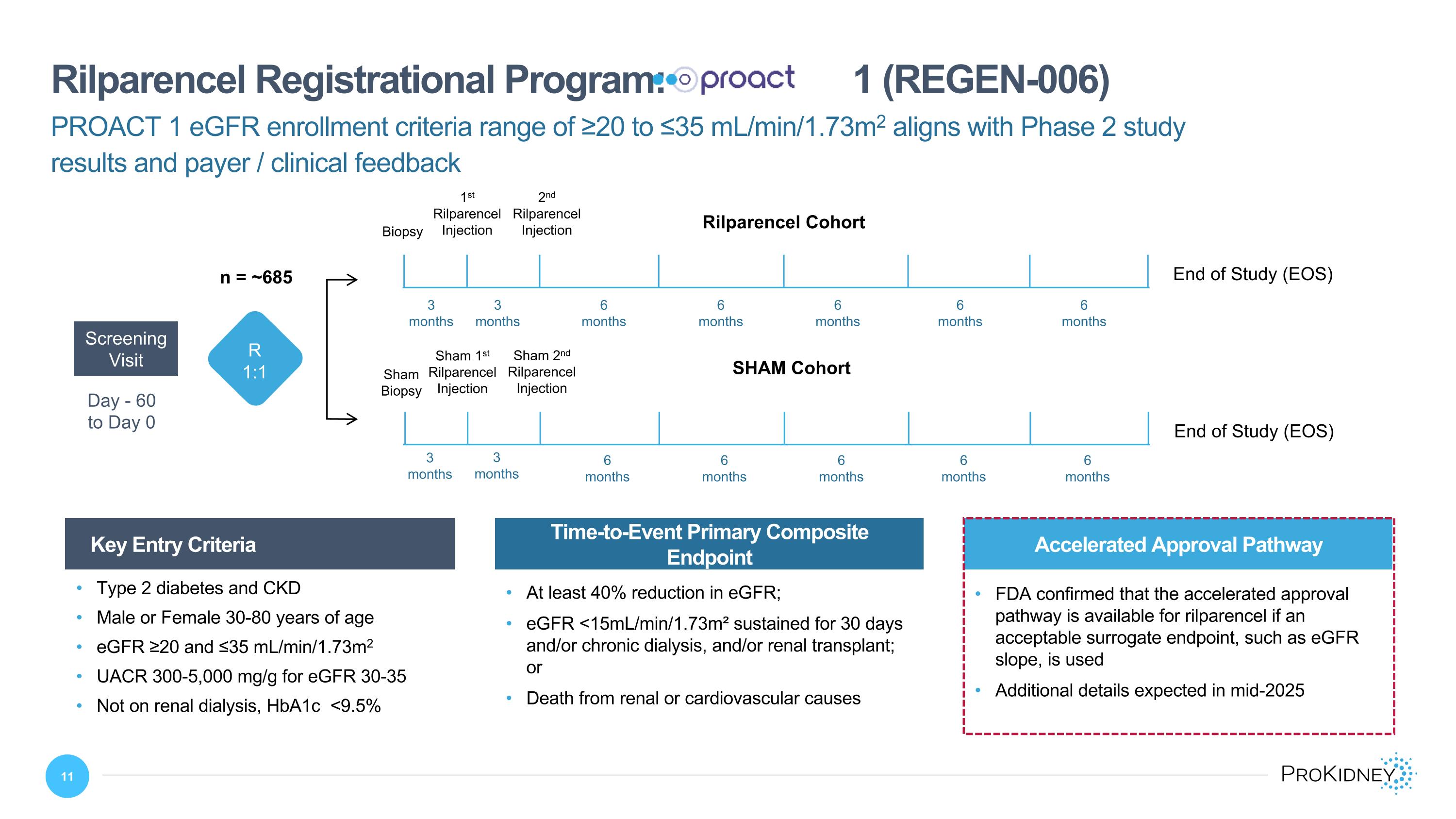
Day - 60 �to Day 0 Screening�Visit SHAM Cohort Sham 1st Rilparencel Injection Sham 2nd Rilparencel Injection Sham Biopsy End of Study (EOS) Rilparencel Cohort 1st Rilparencel Injection 2nd Rilparencel Injection Biopsy End of Study (EOS) PROACT 1 eGFR enrollment criteria range of ≥20 to ≤35 mL/min/1.73m2 aligns with Phase 2 study results and payer / clinical feedback Rilparencel Registrational Program: 1 (REGEN-006) Key Entry Criteria Time-to-Event Primary Composite Endpoint At least 40% reduction in eGFR; eGFR <15mL/min/1.73m² sustained for 30 days and/or chronic dialysis, and/or renal transplant; or Death from renal or cardiovascular causes Type 2 diabetes and CKD Male or Female 30-80 years of age eGFR ≥20 and ≤35 mL/min/1.73m2 UACR 300-5,000 mg/g for eGFR 30-35 Not on renal dialysis, HbA1c <9.5% 3 months 3 months 3 months 3 months 6 months 6 months 6 months 6 months 6 months 6 months 6 months 6 months 6 months 6 months R 1:1 n = ~685 Accelerated Approval Pathway FDA confirmed that the accelerated approval pathway is available for rilparencel if an acceptable surrogate endpoint, such as eGFR slope, is used Additional details expected in mid-2025

REGEN-007 Topline Results
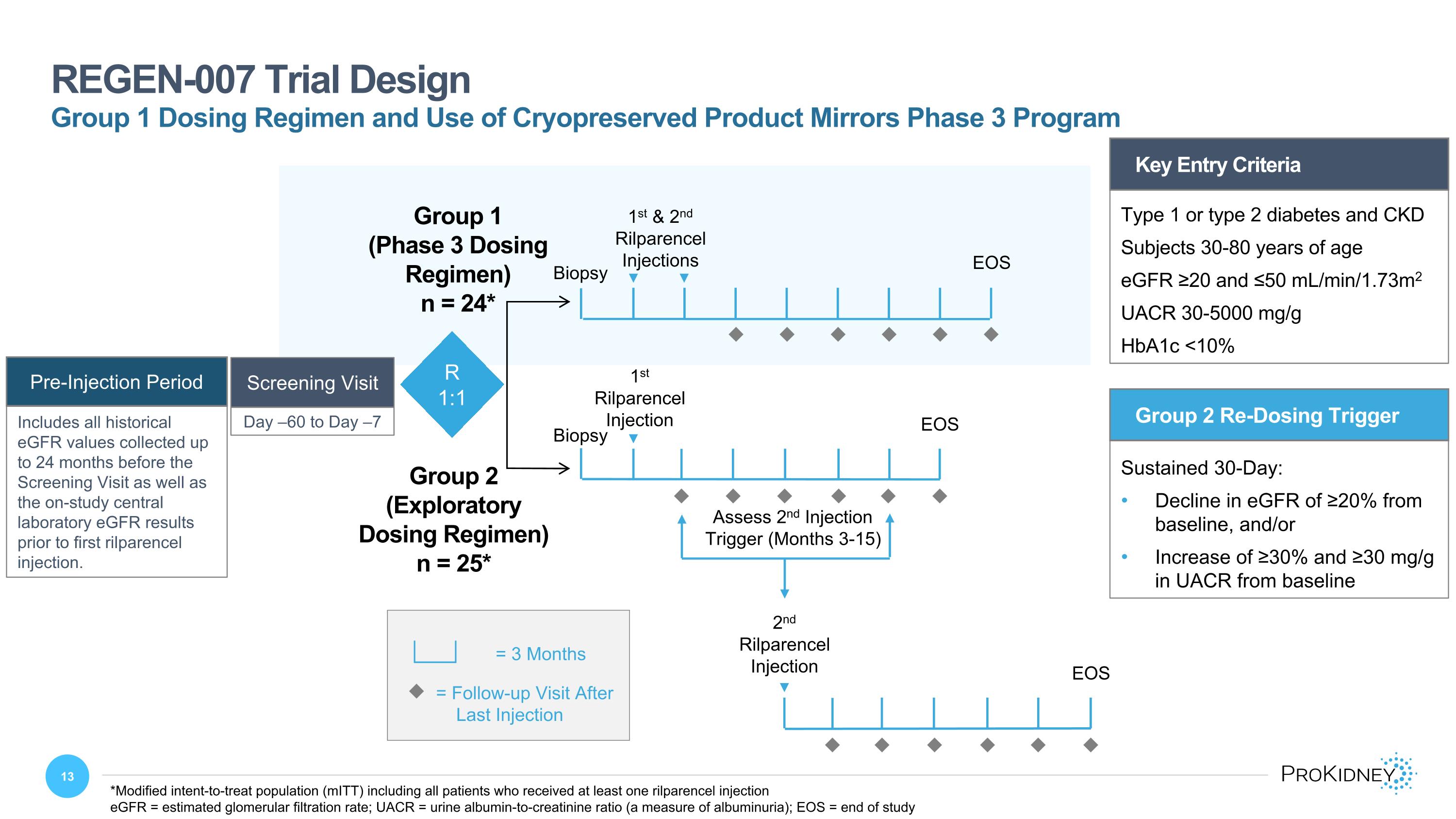
Key Entry Criteria Type 1 or type 2 diabetes and CKD Subjects 30-80 years of age eGFR ≥20 and ≤50 mL/min/1.73m2 UACR 30-5000 mg/g HbA1c <10% Day –60 to Day –7 Screening Visit REGEN-007 Trial Design u = Follow-up Visit After Last Injection = 3 Months Group 2 Re-Dosing Trigger Sustained 30-Day: Decline in eGFR of ≥20% from baseline, and/or Increase of ≥30% and ≥30 mg/g in UACR from baseline 2nd Rilparencel Injection Assess 2nd Injection Trigger (Months 3-15) Group 2 (Exploratory Dosing Regimen) n = 25* R 1:1 1st & 2nd Rilparencel Injections EOS Group 1 (Phase 3 Dosing Regimen) n = 24* EOS 1st Rilparencel Injection u u u u u u u u u u u u u u u u u u EOS *Modified intent-to-treat population (mITT) including all patients who received at least one rilparencel injection eGFR = estimated glomerular filtration rate; UACR = urine albumin-to-creatinine ratio (a measure of albuminuria); EOS = end of study Group 1 Dosing Regimen and Use of Cryopreserved Product Mirrors Phase 3 Program Biopsy Biopsy Pre-Injection Period Includes all historical eGFR values collected up to 24 months before the Screening Visit as well as the on-study central laboratory eGFR results prior to first rilparencel injection.

REGEN-007 Objectives and Endpoints Study Objective Endpoints Evaluate the efficacy, safety, and durability of up to two rilparencel injections on progression of kidney disease in two patient groups using different dosing regimens Primary Efficacy Endpoint Difference in annual eGFR slope in the pre-injection period versus the period following the last rilparencel injection* Primary Safety Endpoint Frequency of procedural and investigational product-related adverse events *Pre-injection period included all historical eGFR values collected up to 24 months before the screening visit as well as the on-study central laboratory eGFR results prior to first rilparencel injection. Period following the last injection included visits from the last rilparencel injection to the EOS visit. Annual eGFR slope calculated using a linear mixed effects model.
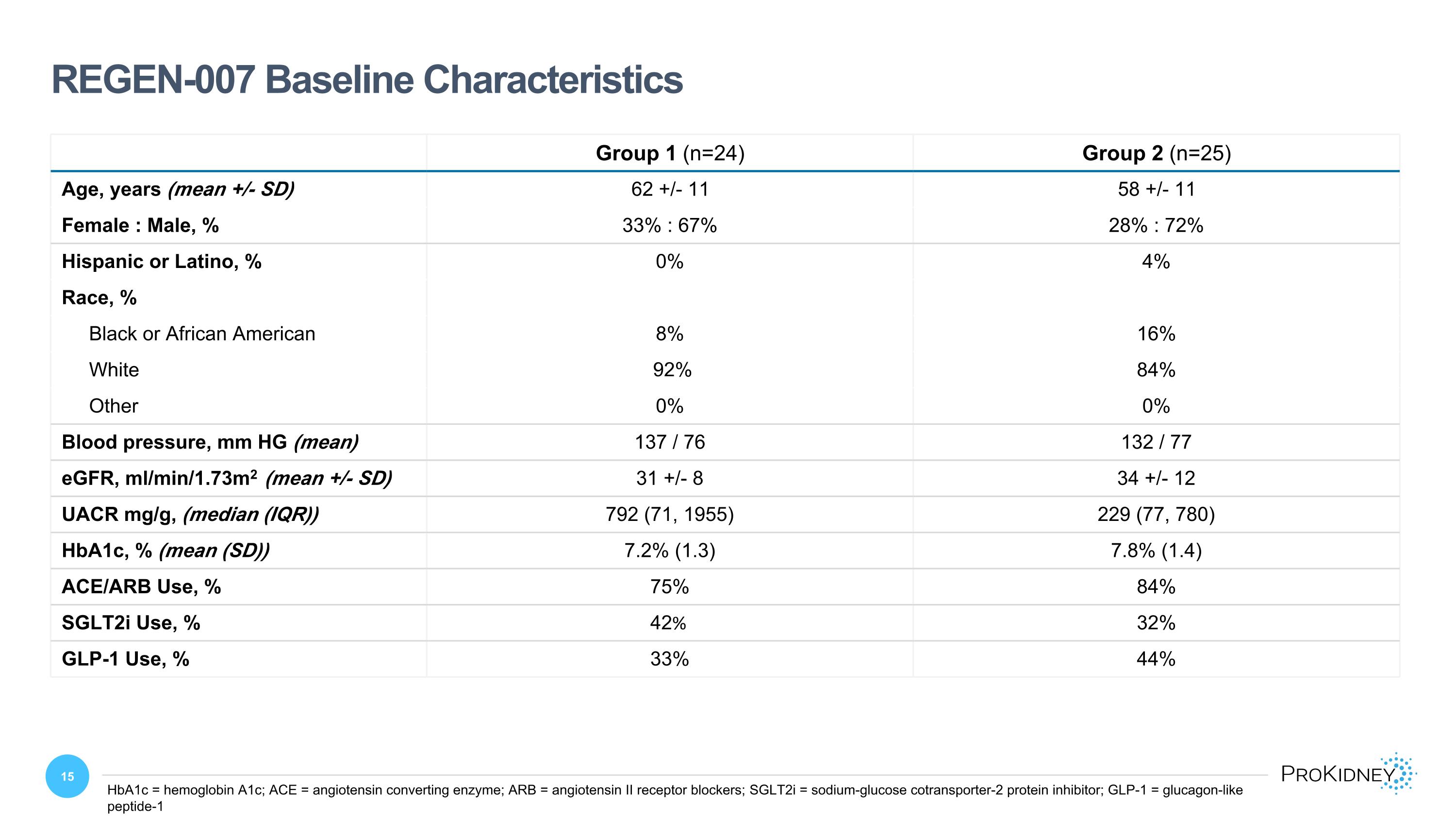
Group 1 (n=24) Group 2 (n=25) Age, years (mean +/- SD) 62 +/- 11 58 +/- 11 Female : Male, % 33% : 67% 28% : 72% Hispanic or Latino, % 0% 4% Race, % Black or African American 8% 16% White 92% 84% Other 0% 0% Blood pressure, mm HG (mean) 137 / 76 132 / 77 eGFR, ml/min/1.73m2 (mean +/- SD) 31 +/- 8 34 +/- 12 UACR mg/g, (median (IQR)) 792 (71, 1955) 229 (77, 780) HbA1c, % (mean (SD)) 7.2% (1.3) 7.8% (1.4) ACE/ARB Use, % 75% 84% SGLT2i Use, % 42% 32% GLP-1 Use, % 33% 44% REGEN-007 Baseline Characteristics HbA1c = hemoglobin A1c; ACE = angiotensin converting enzyme; ARB = angiotensin II receptor blockers; SGLT2i = sodium-glucose cotransporter-2 protein inhibitor; GLP-1 = glucagon-like peptide-1

REGEN-007 Topline Results Group 1 (Phase 3 Dosing Regimen; n=24) Safety (n=49) No rilparencel-related serious adverse events were observed across all patients in the study who received at least one rilparencel injection. The safety profile was consistent with previously reported study results and comparable to a kidney biopsy. Group 2 (Exploratory Dosing Regimen; n=25) Annual decline in eGFR slope* improved by 78% from -5.8 in the pre-injection period to -1.3 in the period following the last rilparencel injection. This 4.6 mL/min/1.73m2 per year difference** was statistically significant (p<0.001) and clinically meaningful. Median follow-up after the last injection was approximately 18 months. Annual decline in eGFR slope* improved by 50% from -3.4 in the pre-injection period to -1.7 in the period following the last rilparencel injection. This 1.7 mL/min/1.73m2 per year difference was not statistically significant (p=0.085) but suggests evidence of a dose response. Median follow-up after the last injection was approximately 18 months. *Annual eGFR slope calculated in mL/min/1.73m2 using a linear mixed effects model **Difference in values is due to rounding
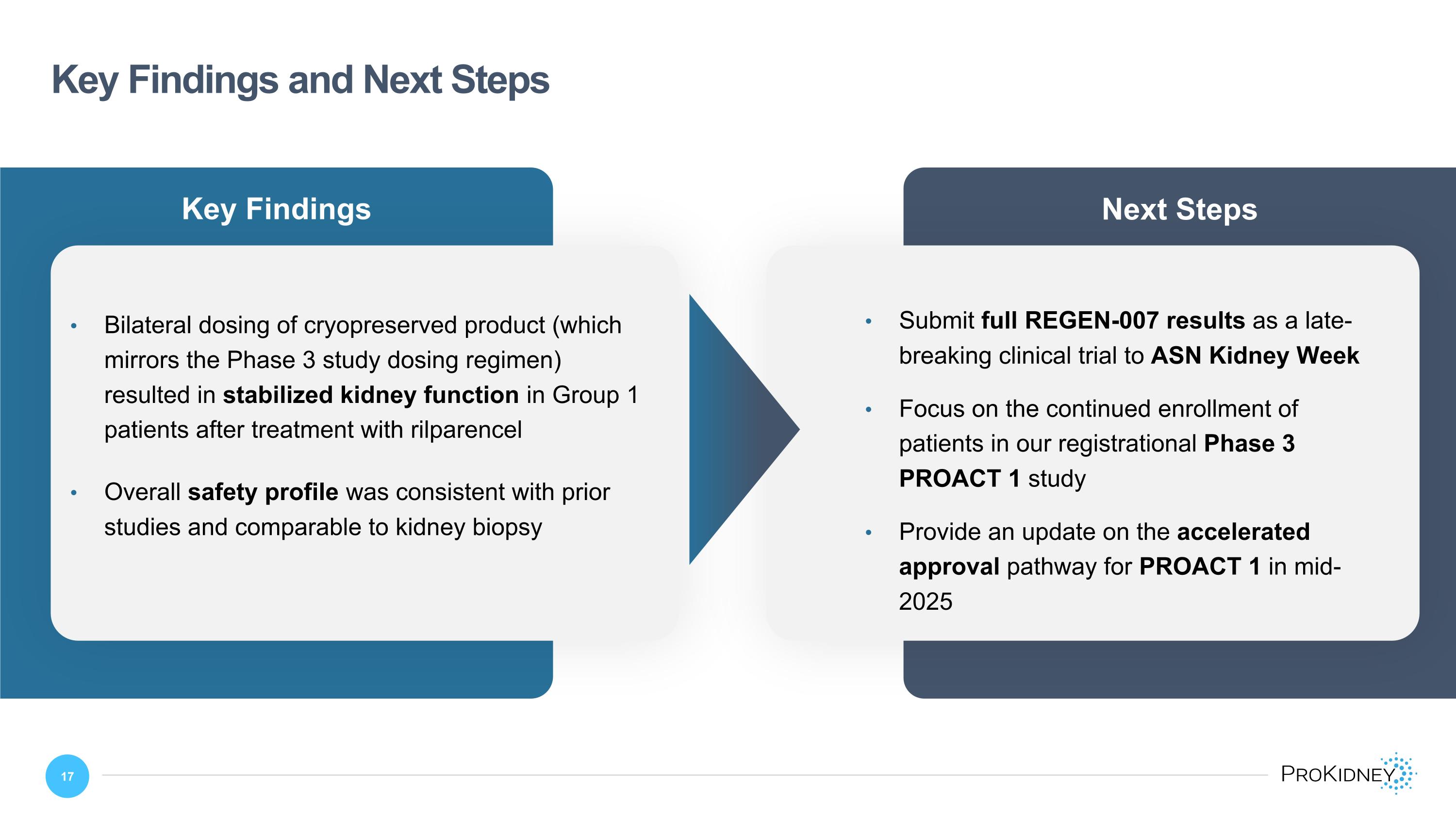
Key Findings Key Findings and Next Steps Bilateral dosing of cryopreserved product (which mirrors the Phase 3 study dosing regimen) resulted in stabilized kidney function in Group 1 patients after treatment with rilparencel Overall safety profile was consistent with prior studies and comparable to kidney biopsy Next Steps Submit full REGEN-007 results as a late-breaking clinical trial to ASN Kidney Week Focus on the continued enrollment of patients in our registrational Phase 3 PROACT 1 study Provide an update on the accelerated approval pathway for PROACT 1 in mid-2025
*As of May 12, 2025 **Cash, cash equivalents and marketable securities as of March 31, 2025 Covering Research Analysts Jason Gerberry Bank of America Yigal Nochomovitz Citi Jonathan Miller Evercore Vamil Divan Guggenheim Kelly Shi Jefferies Anupam Rama JP Morgan Judah Frommer Morgan Stanley Eliana Merle UBS NASDAQ: PROK 292,703,024 shares outstanding* $328M Cash** on hand expected to fund operations into mid-2027 Headquarters: Boston, MA Winston-Salem, NC Financial Highlights




















